FDA Approves New Treatment For ADHD Using Electrical Shocks To The Forehead
NEW YORK (CBSNewYork) – There's an exciting breakthrough for the treatment of ADHD, and it does not involve drugs.
It's the first FDA-approved medical device for the condition and CBS2's Dr. Max Gomez reports that it works by stimulating nerves in the forehead.
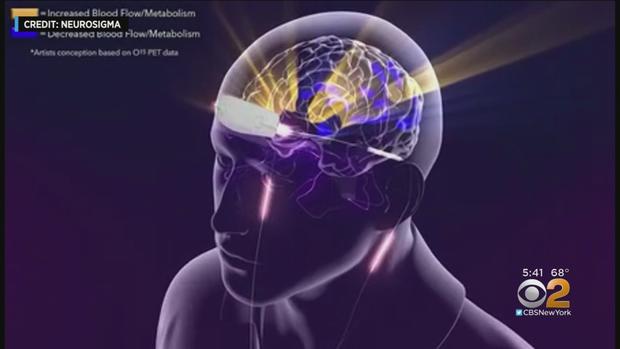
The device, called Monarch eTNS, stimulates different nerves in a similar way that studies have shown stimulating nerves in the neck are effective for controlling epilepsy and depression.
It's easy to think of ADHD as just young kids, usually boys, being full of energy and rambunctious - until you see what one mother deals with when her nine-year-old is unmedicated.
Or what a documentary showed following a young girl with ADHD.
It's a real brain chemistry dysfunction proven by the fact that powerful stimulants used to treat ADHD actually slow these kids down.
Drugs that are essential to helping ADHD children function.
"Untreated, these folks can have other psychiatric problems, including depression, chemical dependency, trouble learning in school, and trouble with the law," said Dr. Jeffrey Borenstein, CEO of the Brain & Behavior Research Foundation.
Since ADHD drugs can have multiple side effects, there's been a search for alternatives.
Now for the first time, a battery powered device offers a non-drug treatment.
Monarch eTNS is a small electrode patch worn on the forehead, usually overnight, that stimulates branches of the sensory trigeminal nerve.
It seems that those low-level electrical impulses travel along the nerves, deep into the brain where they're thought to interact with nearby brain structures - which affect hyperactivity.
Dr. Borenstein says while it's not clear how this affects hyperactivity, since the behavioral brain circuits are complex.
"Those circuits in the brain affecting emotions and behavior can be stimulated with this device and affect the ADHD itself," Dr. Borenstein added.
Double blind studies of the device in 62 children ages seven to 12 found a 44-percent improvement in ADHD symptoms. It's not a cure but it offers a non-drug aid for treatment.
A lot of questions remain. How long will the effect last? Can it be used to lower medication dose? Will it work in older children?
The device from Neurosigma was just approved and is available by prescription for about $1,000. It is not covered by insurance at this point.